CHEM 1010 - INTRO TO CHEMISTRY
MILLER-UREY EXPERIMENT - Origin of Life
STANLEY MILLER

Born in Oakland, California. His parents are Nathan and Edith
Miller from Belarus and Latvia, who had two children. Miller followed his older
brother, attending University of California at Berkeley to study Chemistry. Later,
Miller was offered a teaching assistantship at University of Chicago in
February of 1951. Dr. Miller was recognized for the experiment that was published
in 1953. He devoted the rest of his scientific career at University of
California, San Diego. Spending most of his time researching the well-known research
of simple chemicals being used by living cells that developed during the
geological conditions during early earth’s atmosphere. Stanley Miller died the age of 77 on May 2007.
HAROLD UREY

Harold Clayton Urey was born on April 29 in 1893 – Died January 5,
1981. He was an American Physical Chemist who earned the Nobel Prize in
Chemistry in 1934 for discovering deuterium. He played a noteworthy part in
developing the atom bomb, and his theories on the development of the origin of
life coming from non-living matter. One of his Chicago graduate students was Stanley Miller, who he conducted this unforgettable experiment with.
EARLY EARTH'S ATMOSPHERE
Evidence shows that the Earth’s atmosphere contained much fewer
molecules earlier in time. Major volcanic activity 4 billion years ago
releasing carbon dioxide (CO2), nitrogen (N), hydrogen sulfide (H2O),
and sulfur dioxide (SO2) into the atmosphere. Initially it was
understood that the primitive secondary atmosphere was limited to mainly
ammonia and methane, but scientists also believes the atmosphere correspondingly
had atmospheric carbon that was CO2 with possibly some CO and
nitrogen that was mainly N2. Practicing gas mixtures containing CO,
CO2, N2, etc. would give similar products that contained
CH4 and NH3 as long as O2 wasn't present. In
order to create aromatic amino acids, it would be essential for using less
hydrogen-rich gaseous mixtures. Hydrogen atoms would come from water vapor, and
most of the natural amino acids, pyrimidines, purines, hydroxyacids, and sugars
were formulated in alternative ways during the Miller experiment.
There is abundant evidence of major volcanic eruptions 4 billion years ago, which would have released carbon dioxide (CO2), nitrogen (N2), hydrogen sulfide (H2S), and sulfur dioxide (SO2) into the atmosphere. Experiments using these gases in addition to the ones in the original Miller–Urey experiment have produced more diverse molecules.
There is abundant evidence of major volcanic eruptions 4 billion years ago, which would have released carbon dioxide (CO2), nitrogen (N2), hydrogen sulfide (H2S), and sulfur dioxide (SO2) into the atmosphere. Experiments using these gases in addition to the ones in the original Miller–Urey experiment have produced more diverse molecules.
EXPERIMENT
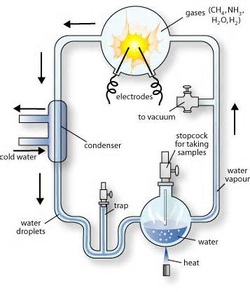
The Miller-Urey experiment was an experiment that replicated the
same atmospheric conditions during early earth and was then tested for the
existence of chemical origins of life. Originally, the experiment was tested
Alexander Oparin’s hypothesis on Earth’s primitive atmosphere that would assist
the chemical reactions that integrated simple organic precursors producing
complex organic compounds. The experimental abiogenesis was accomplished by
Stanley Miller and Harold Urey at the University of Chicago and University of
California, San Diego in 1953 and 1954.
The experiment consisted Water (H2O), Methane (CH4), Ammonia (NH3), and Hydrogen (H2). The chemicals started in a closed sterile glass arrangements of flasks that were linked together in a loop. One flash was half-full of liquid water. Another flask held a pair of electrodes. The liquid water was heated to help it evaporate. They simulated lightning and shocked spark into the electrodes through the atmosphere and water vapor. Once, the atmosphere was cooled again, the water condensed back into a liquid form that eventually dripped back into the first flask and would continue in a cycle.
The experiment mixture had turned into a pink color within a day, Miller and Urey would persist the cycle every two weeks and observed that about 10-15% of Carbon in the system was forming organic compounds. 2% of the carbon had founded amino acids that are used to create proteins in living cells. Sugars were also formed during this experiment, but not nucleic acid during the reaction.
18% of methane molecules converted into bio-molecules.
The rest turned into hydrocarbons like bitumen.
Stanley Miller stated, "Just turning on the spark in a basic pre-biotic experiment will yield 11 out of 20 amino acids."
A professor at UCSD (University of California, San Diego) in the Scripps Institution of Oceanography named Jeffrey Bada, is a former student of Miller and Urey, who has the original experiment remnants under his care. The apparatus is on display at the Denver Museum of Nature and Science.
Scientists have continued to examine Miller’s sealed vials that were conserved from the original experiments, and are able to confirm about 20 additional diverse amino acids than what Miller firstly reported and the twenty that naturally occurs in life. Which makes scientists to believe that maybe the earth’s original atmosphere may have had various compositions than the gases used in the Miller-Urey experiment.
The experiment consisted Water (H2O), Methane (CH4), Ammonia (NH3), and Hydrogen (H2). The chemicals started in a closed sterile glass arrangements of flasks that were linked together in a loop. One flash was half-full of liquid water. Another flask held a pair of electrodes. The liquid water was heated to help it evaporate. They simulated lightning and shocked spark into the electrodes through the atmosphere and water vapor. Once, the atmosphere was cooled again, the water condensed back into a liquid form that eventually dripped back into the first flask and would continue in a cycle.
The experiment mixture had turned into a pink color within a day, Miller and Urey would persist the cycle every two weeks and observed that about 10-15% of Carbon in the system was forming organic compounds. 2% of the carbon had founded amino acids that are used to create proteins in living cells. Sugars were also formed during this experiment, but not nucleic acid during the reaction.
18% of methane molecules converted into bio-molecules.
The rest turned into hydrocarbons like bitumen.
Stanley Miller stated, "Just turning on the spark in a basic pre-biotic experiment will yield 11 out of 20 amino acids."
A professor at UCSD (University of California, San Diego) in the Scripps Institution of Oceanography named Jeffrey Bada, is a former student of Miller and Urey, who has the original experiment remnants under his care. The apparatus is on display at the Denver Museum of Nature and Science.
Scientists have continued to examine Miller’s sealed vials that were conserved from the original experiments, and are able to confirm about 20 additional diverse amino acids than what Miller firstly reported and the twenty that naturally occurs in life. Which makes scientists to believe that maybe the earth’s original atmosphere may have had various compositions than the gases used in the Miller-Urey experiment.
CHEMISTRY OF THE EXPERIMENT
One-step reactions among the mixture components can produce hydrogen cyanide (HCN), formaldehyde (CH2O),and other active intermediate compounds (acetylene, cyanoacetylene, etc.):
CO2 → CO + [O] (atomic oxygen)
CH4 + 2[O] → CH2O + H2O
CO + NH3 → HCN + H2O
CH4 + NH3 → HCN + 3H2 (BMA process)
The formaldehyde, ammonia, and HCN then react by Strecker synthesis to form amino acids and other biomolecules:
CH2O + HCN + NH3 → NH2-CH2-CN + H2O
NH2-CH2-CN + 2H2O → NH3 + NH2-CH2-COOH (glycine)
Furthermore, water and formaldehyde can react, via Butlerov's reaction to produce various sugars like ribose.
The experiments showed that simple organic compounds of building blocks of proteins and other macromolecules can be formed from gases with the addition of energy.
CO2 → CO + [O] (atomic oxygen)
CH4 + 2[O] → CH2O + H2O
CO + NH3 → HCN + H2O
CH4 + NH3 → HCN + 3H2 (BMA process)
The formaldehyde, ammonia, and HCN then react by Strecker synthesis to form amino acids and other biomolecules:
CH2O + HCN + NH3 → NH2-CH2-CN + H2O
NH2-CH2-CN + 2H2O → NH3 + NH2-CH2-COOH (glycine)
Furthermore, water and formaldehyde can react, via Butlerov's reaction to produce various sugars like ribose.
The experiments showed that simple organic compounds of building blocks of proteins and other macromolecules can be formed from gases with the addition of energy.
REFERENCES
"EXOBIOLOGY: An Interview with Stanley L. Miller". Accessexcellence.org.
http://books.nap.edu/openbook.php?record_id=11860&page=85 Exploring Organic Environments in the Solar System (2007)
http://www.britannica.com/EBchecked/topic/382827/Stanley-Miller
http://books.nap.edu/openbook.php?record_id=11860&page=85 Exploring Organic Environments in the Solar System (2007)
http://www.britannica.com/EBchecked/topic/382827/Stanley-Miller

